SYRINGOMYELIA NEWS 2007
 A research update
A research update
By Clare Rusbridge and Penny Knowler
Stone Lion Veterinary Centre, 41 High Street, Wimbledon, London, SW19 5AU
neuro.vet@btinternet.com (CR) Confidential Fax: 020 87860525
penny.knowler@ntlworld.com (SPK)
International Neurology Conference November 2006
This meeting was hosted by the Cavalier Club UK and sponsored by Boehringer Ingelheim Ltd and enabled veterinarians with an interest in syringomyelia to share views and ideas. In addition toinviting veterinarians from Europe, Canada and the USA, the Cavalier Club UK also included breed club representatives from across the UK with the aim of disseminating information aboutthis disease to the wider breeder and dog owning population. Thanks are due to Karlin Lillington for expertise and hard work in reporting the details of the scientific papers and neurologists’ Round Table. These are posted on her website http://sm.cavaliertalk.com and http://www.thecavalierclub.co.uk/ and elsewhere. The outcome of the meeting was very positive and it is hoped that this co-operation between all the specialists and the breeders will result in the research being moved forward. There was agreement on confusing terminology and the revision of the interim breeding guidelines (see websites) The most significant point is that an ‘A’ Grade should only be given to dogs over 2.5 years of age which do not have a syrinx. A hydromyelia is acceptable only if it is less than 2mm in C2/3 area (i.e. barely visible with good resolution MRI).

Neurologist’s round table, International Conference RVC. From the left: Dominic Marino (Long Island Veterinary Specialists),Catherine Louglin (Long Island Veterinary Specialists), James Anderson (Glasgow Veterinary School), Laurent Cauzinille (Centre Hôspitalier Vétérinaire Fregis), Rodolfo Cappello (Royal Veterinary College), Nick Jeffery (Cambridge Veterinary School) Clare Rusbridge (Stone Lion Veterinary Centre), Martin Deutschland (Chestergates Referral), Harvey Carruthers (Stone Lion Veterinary Centre) Graham Flint (Queen Elizabeth Hospital, Birmingham and Ann Conroy Trust), Imelda McGonnell (Royal Veterinary College)
Current Research
 Various
ongoing research programmes are outlined in Clare’s PhD thesis p.192-3. The
successful public defence at Utrecht University on February 15th 2007 was really
a memorable day made much more so by the guard of honour from 20 cavaliers and
the Cavaliergilde Breeders (right).
Various
ongoing research programmes are outlined in Clare’s PhD thesis p.192-3. The
successful public defence at Utrecht University on February 15th 2007 was really
a memorable day made much more so by the guard of honour from 20 cavaliers and
the Cavaliergilde Breeders (right).
The main conclusions of the thesis were as follows:
1) Syringomyelia has a high incidence in the cavalier King Charles spaniel breed and the tendency for it is suspected to be inherited. Preliminary results from a genome scan suggested some interesting regions which warrant further investigation;
2) Syringomyelia is seen in association with a Chiari-like malformation in this breed however a definite link between small caudal fossa volume and fluid cavitation within the spinal cord has yet to be established;
3) It is hypothesised that syringomyelia occurs secondary to cerebrospinal fluid obstruction and abnormal pressure differentials between the spinal cord and subarachnoid space. It is further hypothesised that the syringomyelic fluid is extracellular rather than cerebrospinal in origin;
4) Syringomyelia can result in a neuropathic pain syndrome and this is more likely with a wide syrinx and damage to the spinal cord dorsal horn.
5) Scoliosis is also likely with a wide syrinx and damage to the spinal cord dorsal horn;
6) Medical treatment of syringomyelia associated pain should be directed at agents active at the level of the spinal cord dorsal horn. Drugs that reduce cerebrospinal fluid pressure may also be helpful;
 7)
Surgical cranial cervical decompression can improve clinical signs of pain
however the syringomyelia is generally persistent.
7)
Surgical cranial cervical decompression can improve clinical signs of pain
however the syringomyelia is generally persistent.
A summary information sheet has been included in this newsletter called ‘CM_SM Explained’. Fig 3 –the treatment algorithim has not been updated this time.
Copies of the thesis have been sent to the Chairman/Health Secretaries of the following Clubs/Libraries for members’ use. If there is any other National Club or Institution that would like a copy please contact Penny Knowler.
The Cavalier Club UK, ACKCSC, CKCSC Netherlands, CKCSC of USA, CKCSC Canada, CKCSC Australia, CKSC South Africa (Rand), Association FCKC, CKCS Health Foundation, The American Kennel Club Library, Royal, College Veterinary Surgeons Trust Library, Glasgow Veterinary School Library, Royal Veterinary Collage Library, Carlson Heath Sciences Library (Davis University), University Library Guelph (Canada).
Individuals wanting a personal copy are requested to make a donation to the ‘Syringomyelia DNA research’ fund accessed by PAYPAL and email penny.knowler@ntlworld.com with the address to send the copy. Details http://www.cavalierhealth.org/books.htm or just email Penny.
Genetic Research
We would like to take this opportunity to introduce Dr. Sarah Blott of the Genetics Department at the Animal Health Trust who has joined the CM/SM research team and we are extremely fortunate to have her expertise and experience. Sarah is a geneticist with a particular interest in developing breeding schemes for companion animals that combines state-of-the-art knowledge in quantitative genetics with molecular genetic markers. She has an MSc in Animal Breeding and a PhD in Quantitative Genetics, completed at the Roslin Institute (Edinburgh). Following the award of a Wellcome Trust Travelling Fellowship, she went to the University of Liège, Belgium to work with Professor Michel Georges on methods for fine mapping of complex trait loci in dairy cattle. In 2002 she joined Sygen International plc, one of the world’s leading animal breeding companies, as a Senior Research Scientist and was responsible for data analysis on several research projects in the areas of gene mapping and high throughput gene expression analysis. She has also successfully managed multi-partner European Commission research projects and was a Visiting Scientist at the University of Cambridge Vet School in 2005.
Syringomyelia DNA Research Fund
DNA collection for another genome scan is the most challenging aspect of the research to date. Confidentiality is important because of the reluctance of owners to participate publicly in the project. This particular fund is held by us and is used to
1) disseminate knowledge about the disease and treatment to vets.
2) MRI scanning of particular dogs/family groups (worldwide).
3) facilitate DNA collection.
Buccal swabs have been purchased as an easier means of sending in samples from dogs that blood sampling is not an appropriate option. These can be obtained from Caroline Poulin. Caroline.Poulin@CRCHUM.qc.ca (North America/Australia) or Penny Knowler (Europe/South Africa) penny.knowler@ntlworld.com
Blood (DNA) samples are still needed from MRI confirmed dogs from breeds other than CKCS with/without CM. Please contact Caroline Poulin to arrange shipment.
Donations
We should like to thank all the cavalier lovers for their generous contributions– not only for the specific needs mentioned above but for their moral support. In particular the following people and organisations
• Kendall Barker for a fantastic donation of $450 US for the sale of CKCS
memorabilia on EBay
• Celestial Rainbow Designs for her imaginative enterprise and donation
• Cavalier Club of Canada. $500C has been sent to CHUM for DVD sales
• South African Cavalier Club of the Rand, although a small club, sent a R4386
donation
• Isabelle Barthes and the FCKC a magnificent. $1,048 donation.
2007 International Symposium on Syringomyelia (October 23-26th)
This meeting, taking place at historical setting of Rugby School, England (www.syringomyelia2007.org, email admin@syringomyelia2007.org) is a 3 day event with a veterinary day on the last day (Friday). Abstracts are now being accepted (deadline June 30th and all abstracts will be published in a neurosurgery journal). Deadline for early bird (reduced) registration June 30th 2007. Highlights of the meeting include
• A combined meeting between veterinary and human specialists - a unique
opportunity toshare ideas and experiences
• A dynamic social program including a Ryder Cup course golf tournament for
interested golfers.
• A packed scientific program including surgical approaches, pain management,
genetics, hindbrain, post traumatic and idiopathic syringomyelia, pathology, CSF
physiology and mathematical modeling
• Invited international speakers including Thomas Milhorat, Edward Oldfield,
TatsuyaNagashima and Ulrish Batzdorf.
Rugby is just 1 hour train journey from London in Shakespeare's county and represents the best of English Heritage including the birthplace of the sport of Rugby and the site where the traitors planned the gunpowder plot.
A specific programme for breeders on Friday 26th October is planned (separate “budget” registration fee) but is dependant on whether enough breeders and dog owners are interested (please indicate interest to Clare Rusbridge on neuro.vet@btinternet.com). It is intended to invite the speakers that present on the Veterinary Programme to also give short talks on their current research to the dog owners / breeders.
CANINE CHIARI-LIKE MALFORMATION AND
SYRINGOMYELIA
© Clare Rusbridge BVMS DECVN PhD MRCVS, European and
RCVS Specialist in Neurology
Chiari-like malformation (CM) is a condition characterised by a mismatch between the caudal fossa (skull) volume and its contents, the cerebellum and brainstem. The neural structures are displaced into the foramen magnum obstructing cerebrospinal fluid (CSF) movement. A consequence of this is syringomyelia (SM) where fluid filled cavities develop within the spinal cord (Fig 1). The primary clinical sign of CM/SM is pain, either due to obstruction of the CSF pulse pressure and/or a neuropathic pain syndrome due to damage to the spinal cord dorsal horn. This disease has also been referred to as occipital hypoplasia (Rusbridge et al 2006) and caudal occipital malformation syndrome (COMS) (Dewey et al 2005). CM/SM is sometimes erroneously confused with Arnold Chiari malformation (cerebellar and medulla herniation associated with myelomeningocoele) and occipital dysplasia (incomplete ossification of the supraoccipital bone).
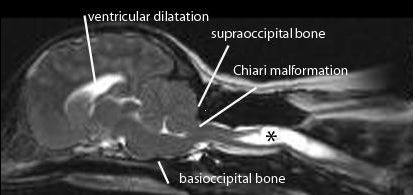 FIGURE
1: Midsagittal T2 weighted MRI of the brain and upper cervical spinal
cord from 3 year old female CKCS with syringomyelia (asterisks) that first
developed signs of pain at 1.7years old. Clinical signs included shoulder
scratching at exercise and when excited. She would not tolerate her right ear to
shoulder area to be touched or groomed. She frequently screamed and her owners
were not able to exercise without her becoming distressed. She also had mild
pelvic limb weakness. She was managed with a foramen magnum decompression and
despite persistence of the syrinx, made a satisfactory post operative that was
maintained for 1.8years. Following this deterioration she was managed medically
for a further 3.8 years and is currently 7 years old.
FIGURE
1: Midsagittal T2 weighted MRI of the brain and upper cervical spinal
cord from 3 year old female CKCS with syringomyelia (asterisks) that first
developed signs of pain at 1.7years old. Clinical signs included shoulder
scratching at exercise and when excited. She would not tolerate her right ear to
shoulder area to be touched or groomed. She frequently screamed and her owners
were not able to exercise without her becoming distressed. She also had mild
pelvic limb weakness. She was managed with a foramen magnum decompression and
despite persistence of the syrinx, made a satisfactory post operative that was
maintained for 1.8years. Following this deterioration she was managed medically
for a further 3.8 years and is currently 7 years old.
PATHOGENESIS
The pathogenesis of canine CM/SM is not fully understood. An important contributory factor is thought to be that the brain is too big for the skull and early studies suggest that there is an inappropriately short skull base (basioccipital bone (Fig 1). Cavalier King Charles spaniels (CKCS) with clinical signs related to syringomyelia are more likely to have a smaller ratio of cauda fossa (i.e. back of skull) volume to total brain volume compared to unaffected CKCS (Cerda-Gonzalez et al 2006). However it is likely there are other unidentified anatomical or environmental factors. Studies comparing skull dimensions did not demonstrate a significant difference between the size of the back of the skull in CKCS with and without syringomyelia (Curruthers et al 2006, Cerda-Gonzalez et al, 2006).
The precise pathogenetic mechanism of development of syringomyelia is much debated (reviewed by Rusbridge et al, 2006; Greiz, 2006). The most popular theory is that obstruction of CSF flow results in relative increase in spinal cord pressure and decrease in pressure in the CSF space around the spinal cord, the consequence of which is repeated mechanical distention of the spinal cord. This in turn results in dilatation of the central canal and accumulation of tissue fluid which eventually coalesces into cavities.
INCIDENCE
The CKCS is overwhelmingly overrepresented for cases of CM/SM. There is no colour or sex predisposition. As shortened skull is a risk factor, any breed with a degree of brachycephalism and/or miniaturization could potentially be predisposed to CM/SM. To date the condition has been also reported in King Charles spaniels, Brussels griffons, Yorkshire terriers, Maltese terriers, Chihuahuas, Miniature dachshunds, Miniature/toy poodles, Bichon Frise, Pugs, Shih Tzus, Pomeranians, Staffordshire bull terriers, a Boston terrier, French bulldogs a Pekingese, a miniature Pinscher and a couple of cats. Recent studies suggest 35% of SM-affected dogs have clinical signs of the condition. The youngest reported dogs with SM have been 12 weeks old. Dogs may be presented at any age although the majority of dogs (approximately 45%) will develop first signs of the disease within the first year of life and approximately 40 % of cases have first signs between 1 and 4 years old. As many as 15% develop signs as mature dogs with the oldest reported case first developing signs of disease aged 6.8 years. Due to the vague nature of signs in some cases and lack of awareness about the disease there is often a considerable time period (mean 1.6 years) between the onset of signs and confirmation of a diagnosis.
CLINICAL SIGNS
The most important and consistent clinical sign of CM/SM is pain however this may be difficult to localise. Owners may describe postural pain; for example, affected dogs may suddenly scream and/or lie with the head on the ground between the paws after jumping up or during excitement. It is also common to sleep with the head in unusual positions, for example elevated. Discomfort often appears worse in the evening and early morning or when excited and can be associated with defecation or may vary with weather conditions. Pain is positively correlated with syrinx width and symmetry (Fig 2); i.e. dogs with a wider asymmetrical syrinx are more likely to experience discomfort, and dogs with a narrow syrinx may be asymptomatic, especially if the syrinx is symmetrical. Dogs with a wide syrinx may also scratch, typically on one side only, while the dog is walking and often without making skin contact, such behaviour is often referred to as an “air guitar” or “phantom” scratching. Dogs with a wide syrinx are also more likely to have scoliosis. In many cases the scoliosis slowly resolves despite persistence of the syrinx.
SM may result in other neurological deficits such as thoracic limb weakness and muscle atrophy (due to ventral horn cell damage) and pelvic limb ataxia and weakness (due to white matter damage or involvement of the lumbar spinal cord by the syrinx). Seizures, facial nerve paralysis and deafness may also be seen; however, no direct relationship has been proven and this association may be circumstantial.
CM alone appears to cause facial pain in some dogs with owners describing ear and facial rubbing/scratching. It has been proposed that CM and compression of the brain stem can result a pain syndrome (Thimineur et al, 2002). In this circumstance it can be difficult to be certain that the CM, as apposed to ear, oral or skin disease, is the cause of the distress especially as CM is a common incidental finding in the CKCS breed.
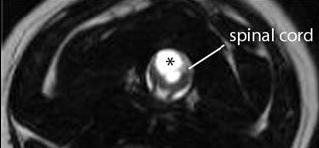 FIGURE
2: T2 weighted transverse image through a wide syrinx (asterisks)
demonstrating the asymmetrical involvement of the right spinal cord dorsalhorn.
FIGURE
2: T2 weighted transverse image through a wide syrinx (asterisks)
demonstrating the asymmetrical involvement of the right spinal cord dorsalhorn.
CLINICAL COURSE
Progression of disease is variable. Some dogs remain stable or deteriorate minimally over years. Other affected dogs can be severely disabled by pain and neurological deficits within 6 months of the first observed signs.
DIAGNOSIS
Magnetic resonance imaging (MRI) is essential for diagnosis and determining the cause of SM (Fig 1). In the instance of CM/SM the cerebellum and medulla extend into or through the foramen magnum which is occluded with little or no CSF around the neural structures. The size of the cerebellar herniation is not correlated with severity. There is typically ventricular dilatation. SM is indicated by fluid-containing cavities within the spinal cord. The upper cervical and upper thoracic segments are typically most severely affected. Maximum syrinx width is the strongest predictor of pain, scratching behaviour and scoliosis; 95% of CKCS with a maximum syrinx width of 0.64cm or more will have associated clinical signs.
CT and radiographs have limited value. In severe cases cervical images may suggest widening of the vertebral canal especially in the C2 region and/or scoliosis. Flexed and extended radiographs of neck can be used to rule out vertebral abnormalities such as atlantoaxial subluxation and for an indication of the likelihood of intervertebral disc disease. Ultrasonography through the cisterna magnum may confirm cerebellar vermis herniation however as CM is so common in the CKCS this information has limited value. Likewise a syrinx may be identified if within the cranial/cervical segment; however, failure to detect a syrinx does not eliminate the possibility of one more caudally. CM/SM does not appear to increase risk of anaesthesia.
DIFFERENTIAL DIAGNOSIS
The most important differential diagnoses are other causes of pain and spinal cord dysfunction such as intervertebral disc disease; CNS inflammatory diseases such as granulomatous meningoencephalomyelitis; vertebral abnormities such as atlantoaxial subluxation; neoplasia; and discospondylitis. When scratching or facial/ear rubbing is thepredominant clinical sign, ear and skin disease should be ruled out. The scratching behaviour for SM is classically to one distinct area. It is a common incidental finding for CKCS to have a mucoid material in one or both tympanic bullae and in the majority of cases this is not associated with clinical signs. Some cases with scoliosis appear to have a head tilt which could be confused with vestibular dysfunction. If in doubt cervical radiographs can confirm scoliosis.
TREATMENT
The main treatment objective is pain relief. The most common surgical management is cranial/cervical decompression (also described as foramen magnum or suboccipital decompression) establishing a CSF pathway via the removal of part of the supraoccipital bone and dorsal arch of C1. This may be combined with a durotomy (incision of the dura with/without incision of subarachnoid meninges) with or without patching with a suitable graft material. Cranial/cervical decompression surgery is successful in reducing pain and improving neurological deficits in approximately 80% of cases and approximately 45% of cases may still have a satisfactory quality of life 2 years postoperatively (Rusbridge 2007). However surgery may not adequately address the factors leading to SM and the syrinx appears persistent in many cases (Rusbridge 2007). The clinical improvement is probably attributable to improvement in CSF flow through the foramen magnum. In some cases scaring and fibrous tissue adhesions over the foramen magnum seem to result in re-obstruction and 25% to as many as 50% of cases can eventually deteriorate (Dewey et al 2005, Rusbridge 2007). This can be as early as 2 months postoperatively. Recently, a cranioplasty procedure used in human cranial/cervical decompression surgery has been adapted for use in dogs. The procedure entails placement of a plate constructed of titanium mesh and polymethylmethacrylate (PMMA) on pre-placed titanium screws bordering the occipital bone defect (Dewey et al 2006). An alternative method of managing SM is direct shunting of the cavity. In humans this is not a preferred technique as long term outcome is poor due to shunt obstruction and/or spinal cord tethering. There has been a single report of syringo-subarachnoid shunting in a dog using an equine ocular lavage tube. However post-operative MRI revealed that SM was still prominent although there was a clinical improvement in the dog (Skerritt and Hughes 1998).
Due to the persistence of SM and/or spinal cord dorsal horn damage it is likely that the post-operative patient will also require continuing medical management for pain relief and in some patients medical management alone is chosen because of financial reasons or owner preference. There are three main drugs used for treatment of CM/SM: drugs that reduce CSF production; analgesics; and corticosteroids (Fig 3). If the dog’s history suggests postural pain or discomfort relating to obstruction of CSF flow then a trial of a drug which reducing CSF pressure, e.g. furosemide, cimetidine or omeprazole, is appropriate. This can also be very useful if it is difficult to determine if the cause of discomfort is CM versus, for example, ear disease. CSF pressure reducing drugs may be sufficient to control signs in some dogs, but additional analgesics are likely to be necessary for an individual with a wide syrinx. In this circumstance we suggest that non steroidal anti-inflammatory drugs are the medication of first choice partly because there are several licensed products. However, for dogs with signs of neuropathic pain, i.e. allodynia and scratching behaviour (suspected dysesthesia); a drug which is active in the spinal cord dorsal horn is more likely to be effective. Because gabapentin has established use in veterinary medicine we suggest that this is the drug of first choice but amitriptyline or pregabalin may also be suitable. Corticosteroids are an option if pain persists or where available finances prohibit the use of other drugs. Because the mechanisms of development of neuropathic pain are multifactorial, appropriate polypharmacy is likely to be more effective than treatment with single agents. Anecdotally, acupuncture and ultrasonic treatments have been reported to be useful adjunctive therapy in some cases. The dog’sactivity need not to be restricted but owner should understand that dog may avoid some activities and grooming may not be tolerated. Simple actions, for example raising the food bowl and removing neck collars, can also help.
PROGNOSIS
Prognosis for CM/SM managed medically is guarded especially for dogs with a wide syrinx and/or with first clinical signs before 4 years of age. Study of a small case series (14 CKCS) managed conservatively for neuropathic pain suggested that 36% were eventually euthanatized as a consequence of uncontrolled pain. However 43% of the group survived to be greater than 9 years of age (average life expectancy for a CKCS is 10.7 years). Most dogs retain the ability to walk although some may be significantly tetraparetic and ataxic.
BREEDING RECOMMENDATIONS
Current breeding recommendations for CKCS concentrate on removal of dogs with early onset SM (i.e. within the first 2.5 years of life) from the breeding pool (for precise recommendations and grading system see http://www.cavaliertalk.com/SM).
REFERENCES AND FURTHER READING
Carruthers H, Rusbridge C, Dubé, M-P, et al "Association between cervical and intracranial dimensions and syringomyelia in the cavalier King Charles spaniel" In: Rusbridge C, Chiari-like malformation and Syringomyelia in the Cavalier King Charles spaniel. ISBN 90-393-4456-6; 978-90-393-4456-9. 82, 2006
Cerda-Gonzalez S, Olby NJ, Pease TP: Morphology of the Caudal Fossa in Cavalier King Charles Spaniels. J Vet Internal Med 20: 736, 2006
Dewey CW, Bailey KS, Marino DJ, et al. Foramen magnum decompression with cranioplasty for treatment of caudal occipital malformationsyndrome in dogs.
Dewey CW, Berg JM, Barone G et al: Foramen magnum decompression for treatment of caudal occipital malformation syndrome in dogs. J Am Vet Med Assoc 227: 1270, 2005
Greitz D: Unravelling the riddle of syringomyelia. Neurosurg Rev 29:251, 2006
Levine DN: The pathogenesis of syringomyelia associated with lesions at the foramen magnum: a critical review of existing theories and proposal of a new hypothesis. J Neurol Sci; 220:3, 2004.
Lillington, K, http://www.cavaliertalk.com/SM - a resource for current information on treatment and breeding recommendations with links to Chiari-like malformation and syringomyelia discussion and support groups.
Rusbridge C, Greitz D, Iskandar BJ: Syringomyelia: Current concepts in pathogenesis, diagnosis and treatment. J Vet Internal Med 20: 469, 2006
Rusbridge C, Jeffery, NJ: Pain mechanisms and treatment in Chiari malformation and syringomyelia in the dog. In press The Veterinary Journal (2007), doi:10.1016/j.tvjl.2006.12.007.
Rusbridge C, Knowler SP: Inheritance of occipital bone hypoplasia (Chiari type I malformation) in cavalier King Charles spaniels. J Vet Internal Med; 18:673, 2004.
Rusbridge C: Chiari-like malformation with syringomyelia in the cavalier King Charles spaniel; long term follow up after surgical management. In press Veterinary Surgery.
Skerritt GC, Hughes D: "A syndrome of syringomyelia in the cavalier King Charles spaniel, and its treatment by syringo-subarachnoid shunting." In: Proceedings from the 12th Annual Symposium of the European Society of Veterinary Neurology, Vienna, 23: 1998.
Smith SR. For the Love of Ollie; a story of compassion and courage. Trimatrix Management Consulting Inc, 2047 Pen Street, Oakville, Ontario, Canada, available though http://www.fortheloveofollie.com, 2006. This book is a valuable resource for owners and their veterinarians and was written by the owner of 2 CM/SM affected dogs. It describes her journey from first realising that her pet was in pain to the post-operative period. There are contributions from other CM/SM affected dog owners and a simple explanation of the disease and treatment.
Thimineur M, Kitaj M, Kravitz E et al: Functional abnormalities of the cervical cord and lower medulla and their effect on pain: observations in chronic pain patients with incidental mild Chiari I malformation and moderate to severe cervical cord compression. Clin J. Pain 18:171, 2002
Related Links
Syringomyelia
SM
Breeding Protocol
Dr. Rusbridge's Syringomyelia News Winter 2007 Research Update
Dr. Rusbridge's Syringomyelia News Autumn 2007 Research Update
Board Certified Veterinary Neurologists
Questions for Cavalier Breeders
MRI Screening Protocol
